Many reactions emit energy, often in large amounts, but cosmic efficiency is another metric altogether. Here’s how to maximize your output.
In terms of making things happen, energy is an indispensable consideration.

Systems spontaneously tend towards the lowest-energy state.

When a system reaches equilibrium, no further energy can be extracted.

That maximum entropy, lowest energy state is the inevitable end-state of the Universe.

But until that moment arrives, reactions of all kinds will occur, continuing to liberate energy.
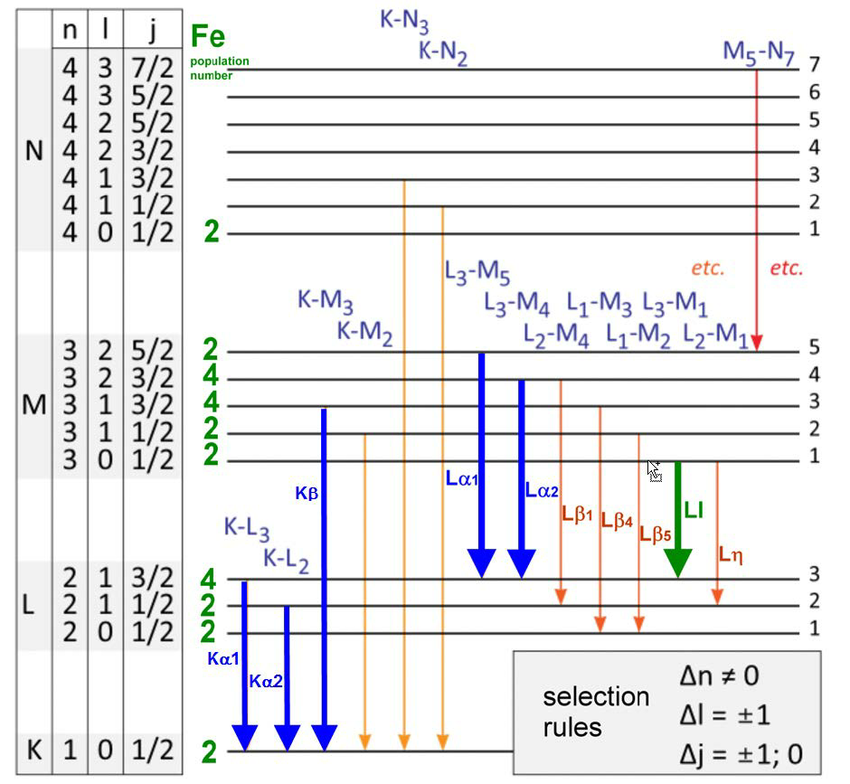
In our bodies, chemical bonds break and reform: releasing energy.
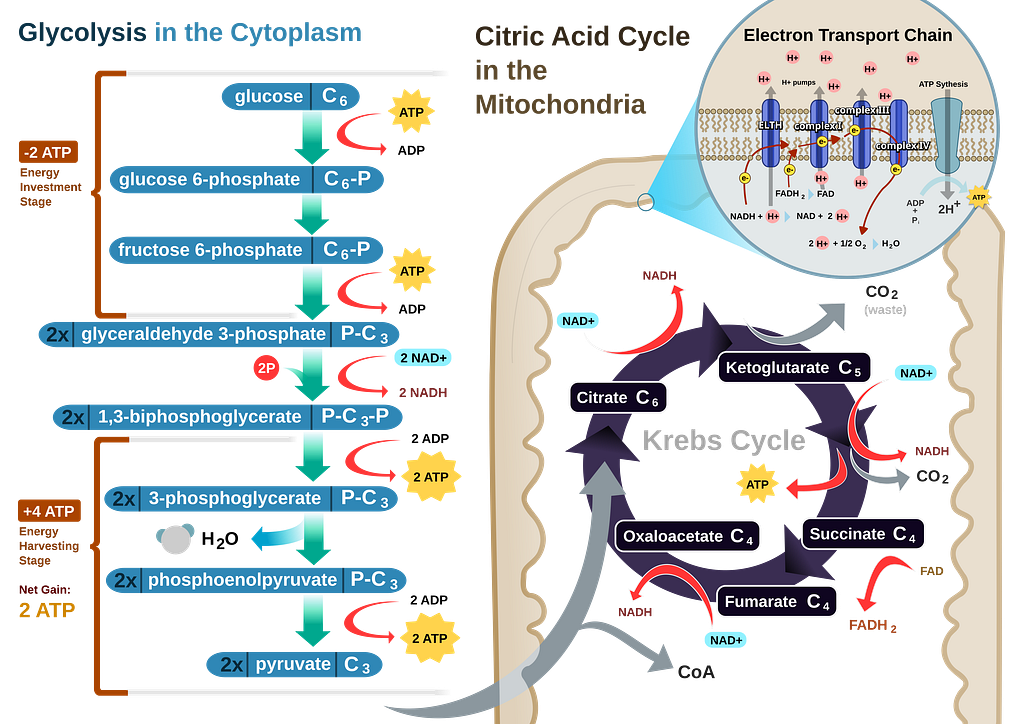
Aerobic respiration releases 2.88 megajoules of energy per mole of sugar.

Only 0.0000000094% of the initial fuel’s mass converts into energy.
Chemical reactions are more efficient, like combustion.

TNT’s heat of combustion is 14.5 megajoules per kilogram: just 0.000000016% efficient.

Rocket fuels, like RP-1 and mixing liquid hydrogen/oxygen, are only ~10 times more efficient.

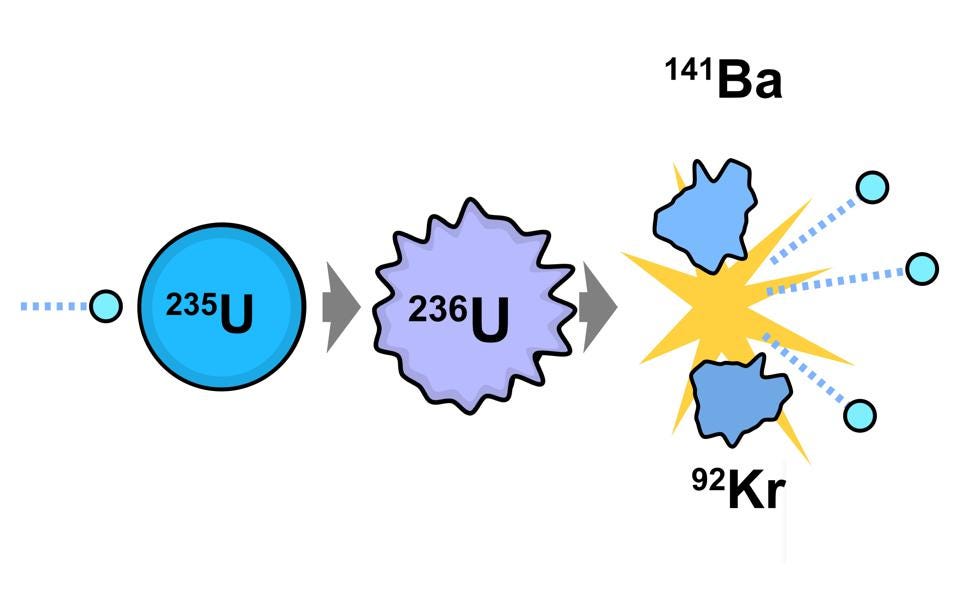
When enriched uranium undergoes nuclear fission, efficiencies are much higher.

Each U-235 kilogram liberates 72 trillion joules: 0.08% efficient.
Nuclear fusion, like in the Sun, liberates 630 trillion joules for each kilogram of hydrogen fuel.

At 0.7% efficiency, it’s the holy grail of our clean energy ambitions.

Stars use fusion to generate hundreds of times the energy stored even gravitationally.

Only matter-antimatter annihilation is more energy efficient.

Mass converts 100% to energy via E = mc²: the perfect fuel source.

Mostly Mute Monday tells an astronomical story in images, visuals, and no more than 200 words.
Starts With A Bang is written by Ethan Siegel, Ph.D., author of (affiliate links following) Beyond The Galaxy, Treknology, The Littlest Girl Goes Inside An Atom, and Infinite Cosmos. His latest, The Grand Cosmic Story, is out now!
What are the most energy-efficient reactions in physics? was originally published in Starts With A Bang! on Medium, where people are continuing the conversation by highlighting and responding to this story.